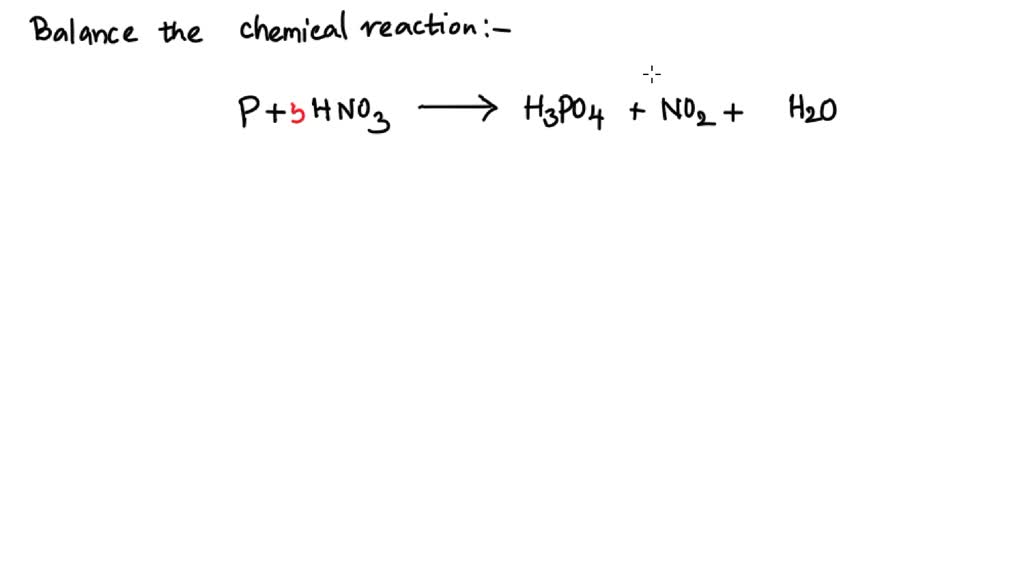
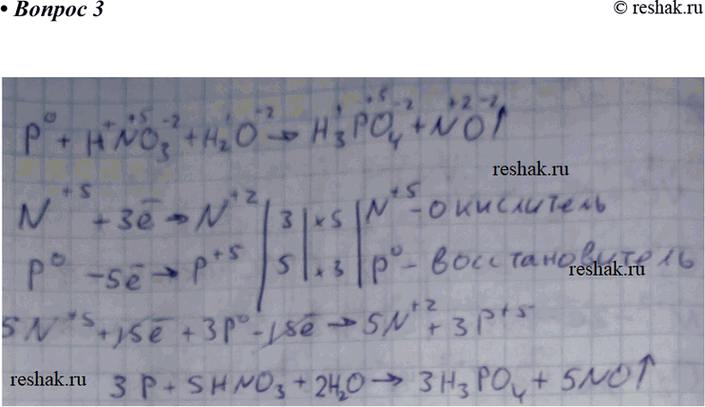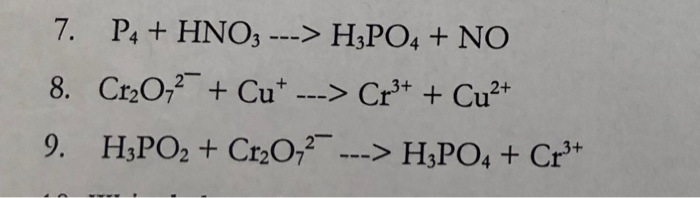Concentrated nitric acid oxidises phosphorus to phosphoric acid according to the following equation: P +5HNO3 → H3PO4 +5NO2+ H2O If 6.2 g of phosphorus was used in the reaction, calculate: a The

Oxidation Number method. P4+HNO3+H2O=H3PO4+NO. Balance the equation by oxidation Number method. - YouTube